- PainChek is expanding its namesake pain-assessment and monitoring app in a partnership with Murdoch Children’s Research Institute (MCRI)
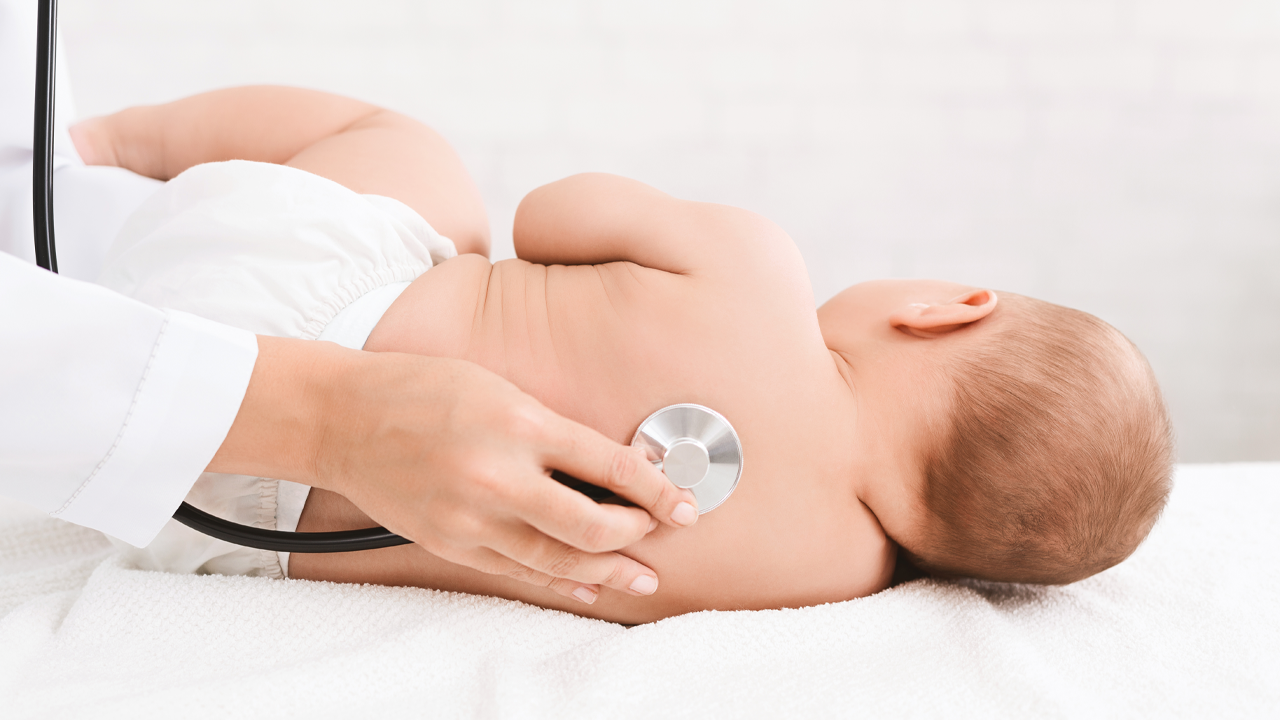
- A study conducted through the partnership will test a new infant version of the PainChek mobile app on 100 infants in the Emergency Department of the Royal Children’s Hospital in Melbourne
- The PainFace study is planned to commence “within days”
- Results from the clinical study will help PainChek gain regulatory approval for the infant version of its app in Europe and the USA
PainChek is partnering with Murdoch Children’s Research Institute (MCRI) to expand its pain-assessment mobile app by testing it on infants.
The PainChek Mobile App is a pain-assessment and monitoring program designed for people who can’t communicate their pain, such as sufferers of dementia. The mobile-based app takes a short video of a patient’s face using a smart phone camera and then uses facial recognition analytics to recognise facial muscle movements that indicate pain.
A doctor or caregiver can then record observations of pain-related behaviours, like how the patient is moving or vocalising pain. PainChek then calculates and stores an overall pain score, helping caregivers monitor the effect of medication and treatment over time.
The app was initially designed with dementia-sufferers in mind but is now expanding to an infant-specific product with the help of MCRI. A clinical study will use the infant-focussed version of the PainChek app to compare pain scores with an observational children’s pain assessment tool known as the FLACC (Face, Legs, Activity, Cries and Consolability) scale.
The PainFace study, which is planned to commence “within days”, will test roughly 100 infants undergoing painful procedures in the Emergency Department (ED) of the Royal Children’s Hospital in Melbourne.
According to PainChek, MCRI is the largest child health research institute in Australia, and in the top three globally.
PainChek Chief Scientific Officer Professor Jeff Hughes said the company was excited to work with researchers from MCRI.
“Children who present to the ED often have underlying pain or need to go through painful medical procedures. The PainFace study will be looking to validate PainChek as an accurate pain assessment tool that helps clinicians in the ED improve the quality of pain management for these vulnerable children,” Jeff said.
Results from the clinical study will help PainChek gain regulatory approval for the infant version of its app in Europe and the USA.
PainChek shares are currently up 2.5 per cent, trading at 20 cents each in a $185.87 million market cap as at 12:26pm AEST today.