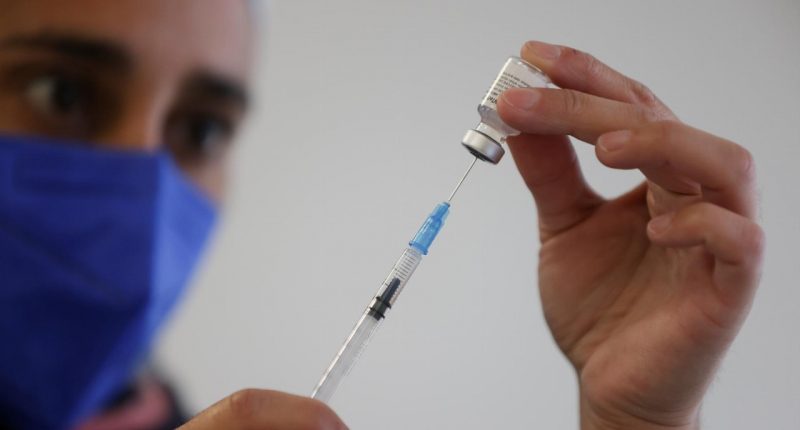
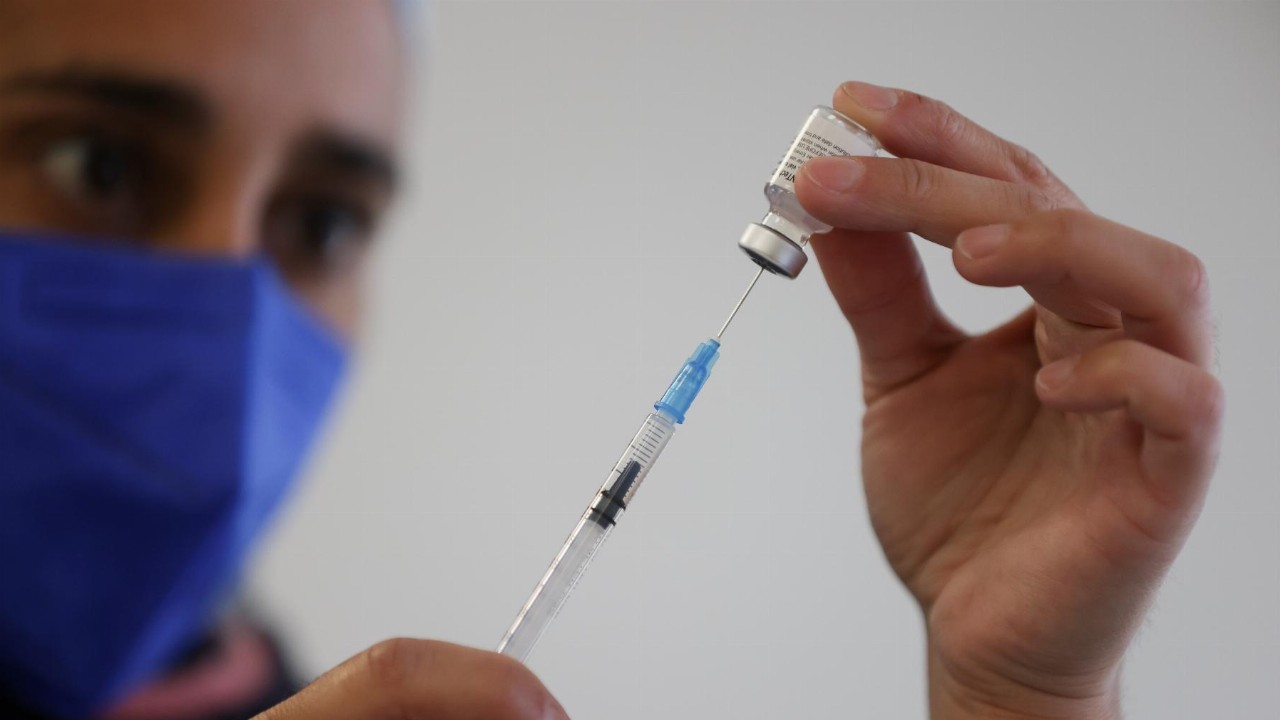
- Pfizer said on Thursday it would ask US regulators to authorise a booster dose of its vaccine within the next month
- Based on Pfizer’s own studies, a third booster shot generates antibody levels that are five- to 10-times higher than after the second dose
- The company said it has already been approached by several European countries to discuss booster doses
- However, some experts say basing the decision on waning antibody protection ignores the role of important other parts of the immune response
- Pfizer intends to launch a placebo-controlled trial of the booster with 10,000 participants
With the highly infectious Delta strain of COVID-19 continuing to spread, Pfizer said on Thursday it would ask US regulators to authorise a booster dose of its vaccine within the next month.
The drugmaker’s Chief Scientific Officer Mikael Dolsten cited a recently reported drop in the shot’s effectiveness in Israel, saying it was mostly due to infections in people who had been vaccinated in January or February. According to the country’s health ministry, the ability of Pfizer’s vaccine to prevent both infection and symptomatic disease fell to 64 per cent in June.
“The Pfizer vaccine is highly active against the Delta variant,” Dolsten said in an interview. But after six months, he added, “there likely is the risk of reinfection as antibodies, as predicted, wane.”
“It’s a small data set, but I think the trend is accurate: six months out, given that Delta is the most contagious variant we have seen, it can cause infections and mild disease.”
Still, Dolsten stressed that the data from Israel, as well as from Britain, suggests that the vaccine is 95 per cent effective against severe disease, even with the drop in antibodies.
Based on information from Pfizer’s own studies, a third booster shot generates antibody levels that are five to 10-times higher than after the second dose, which may be particularly important for older people.
Dolsten said several European countries had already approached Pfizer to discuss booster doses and that some may start administering them even before US authorisation.
However, some experts have raised concerns. Dr Eric Topol, a professor of molecular medicine and director of the Scripps Research Translational Institute in California, said basing the decision on waning antibody protection ignores the role of important other parts of the immune response, including memory B cells, which are capable of producing antibodies on demand when challenged by the virus.
“You need better studies to be able to assert that. It isn’t just neutralising antibodies,” Topol said.
Pfizer intends to launch a placebo-controlled trial of the booster with 10,000 participants. The study is expected to continue for several months, meaning it will not be completed in time for the company’s filing with the US Food and Drug Administration.
But even if Pfizer succeeds in securing FDA approval, it’s just the first step. The booster will. still need to be reviewed and recommended by advisers to the US Centres for Disease Control and Prevention.
“It’s not automatic by any means,” said Dr William Schaffner, a vaccine expert at Vanderbilt University.