- ResApp Health (RAP) will soon launch SleepCheck, an at-home obstructive sleep apnoea (OSA) screening phone app
- SleepCheck is a simple but clinically accurate app that assesses a person’s risk of OSA
- It does this by analysing their breathing patterns during sleep
- OSA is the most common sleep disorder and, when undiagnosed, can lead to severe problems such as heart disease, hypertension or a stroke
- Unfortunately, studies have shown that around 80 per cent of people with OSA are undiagnosed
- The company hopes SleepCheck will lower this statistic once it launches this quarter
- ResApp is up 7.69 per cent and shares are trading for 21 cents each
ResApp Health (RAP) will soon launch SleepCheck, an at-home obstructive sleep apnoea (OSA) screening smartphone app.
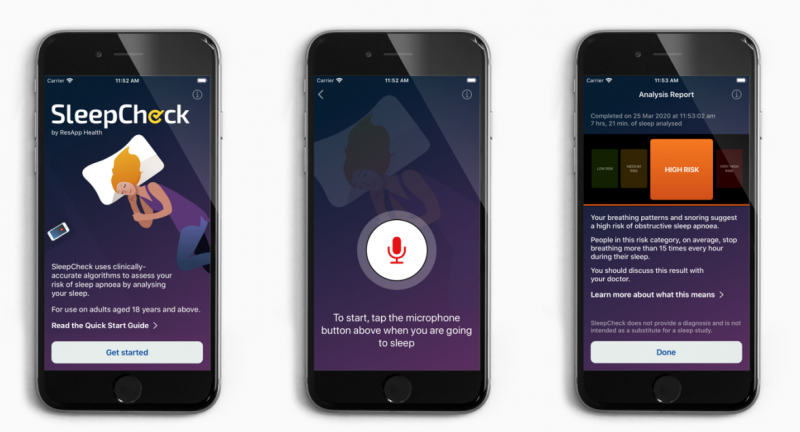
SleepCheck is supposedly easy to use and offers clinically accurate algorithms to assess a person’s risk of OSA. It does this by analysing their breathing and snoring sounds during sleep.
SleepCheck doesn’t require any additional accessories or hardware other than the user’s smartphone.
Sleep apnoea affects more than three in every 10 men and nearly two in every 10 women. Sleep apnoea is when the air stops flowing to your lungs for 10 seconds or longer during sleep, thus creating an ‘apnoea’ event.
Studies have shown that 80 per cent of people with sleep apnoea are undiagnosed. If left untreated, sleep apnoea can lead to heart disease, hypertension, stroke and type 2 diabetes.
“Undiagnosed sleep apnoea causes poor sleep and daytime tiredness, and can have a major impact on a person’s health,” CEO and Managing Director Tony Keating said.
“We are excited to announce that SleepCheck will soon be available to download on the App Store,” Tony said.
Initially, the app will be launched in Australia and the U.K, with pricing still to be determined.
In a clinical study, ResApp’s algorithms identified 238 patients with OSA of all sensitivities. This compared to an American Academy of Sleep Medicine Type II sleep study.
“It is backed by a large clinical study which demonstrated that the SleepCheck algorithms were able to accurately identify OSA when compared to a full at-home comprehensive sleep study,” he continued.
“SleepCheck fills a unique and very important place in the evaluation of OSA between questionnaire-based screening and use of increasingly complex types of sleep studies requiring multiple wires and sensors,” Cardio Respiratory Sleep’s cardiologist Dr Philip Currie said.
SleepCheck is CE marked and will be listed on the Australian Register of Therapeutic Goods shortly.
ResApp is up 7.69 per cent and shares are trading for 21 cents each at 11:01 am AEDT.







