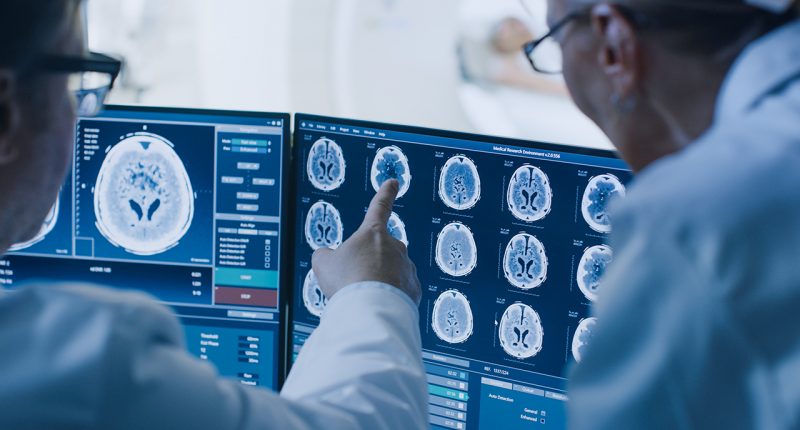
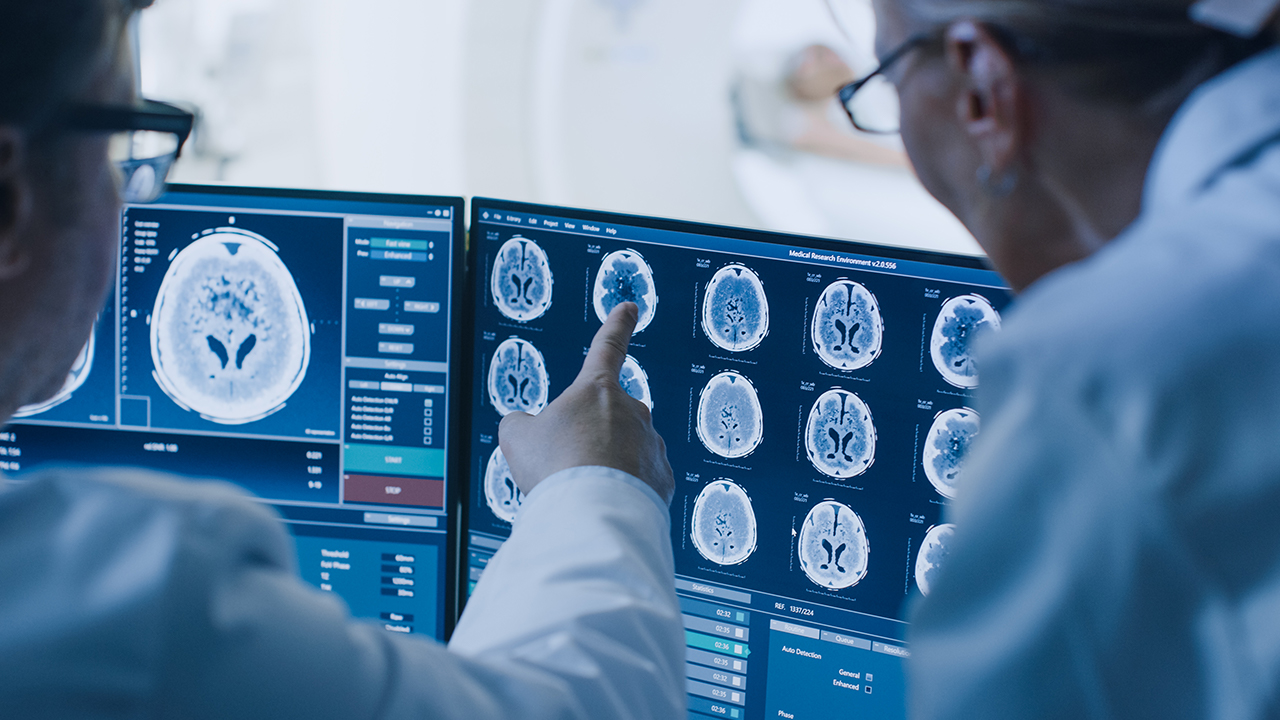
- Resonance Health (RHT) has been contracted to provide liver-fat quantification services for a clinical study to treat non-alcoholic Steatohepatitis
- This contract marks the first time the company’s HepaFat-AI medical device will be used in a clinical study environment
- The study is expected to span 18 months and if Resonance withstands this duration, the contract value is estimated to be $100,000
- Resonance says this trial validates its medical device and establishes a precedent for other clinical trial procurement efforts
- At market close shares were up 3.2 per cent to trade at 16 cents
Resonance Health (RHT) has been contracted to provide liver-fat quantification services for a clinical study to treat non-alcoholic Steatohepatitis (NASH).
Invictus Ops, a wholly owned subsidiary of NSX-listed VGI Health Technology, is undertaking the NASH Phase II clinical study which is expected to span 18 months and involve approximately 100 trial participants.
The contract marks the first time Resonance Health’s recently regulatory-cleared HepaFatAI medical device will be used in a clinical study environment to measure, grade and quantify the liver fat of the trial participants at two to three timepoints over the 18-month period.
The clinical study is due to commence recruitment of patients shortly and VGI Managing Director Dr Glenn Tong said the partnership with Resonance Health will “vastly improve” the efficiency in which the key end points are measured.
Fatty liver disease is reportedly emerging as a major global health issue and, according to the company, is attracting significant attention from international pharmaceutical companies seeking to develop effective drug treatments for the disease.
Resonance Health said participating in this trial validates its medical device and establishes a precedent for other clinical trial procurement efforts.
The contract value is estimated to be approximately $100,000. Under the terms of the agreement, however, VGI may discontinue or extend the study at any time in which case Resonance would receive payment for the services performed up to the termination date.
With fatty liver diseases on the rise, Resonance Health Managing Director Mitchell Wells said the company is delighted to assist Invictus and applauds its efforts to find treatments for NASH.
“Resonance Health can help combat this global epidemic with its products and services that quantify and measure fat in the liver,” Mr Wells said.
“The study is a material achievement in that this is the first time HepaFat-AI will be
used in a NASH clinical trial, and it sets an important precedent.”
At market close on Tuesday, shares in Resonance Health were up 3.2 per cent to trade at 16 cents.