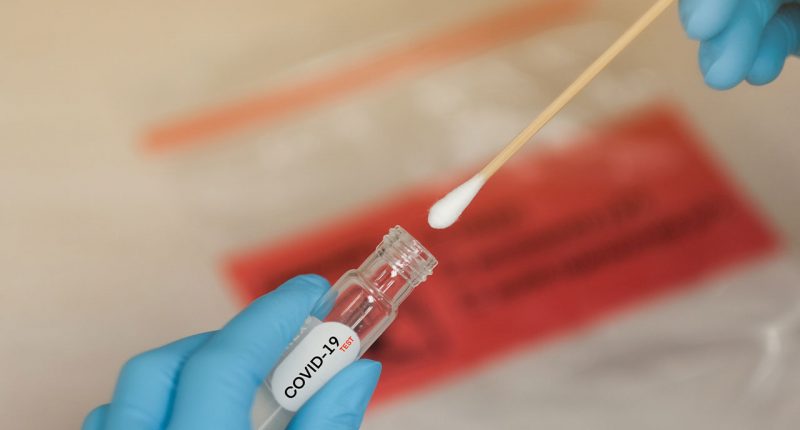
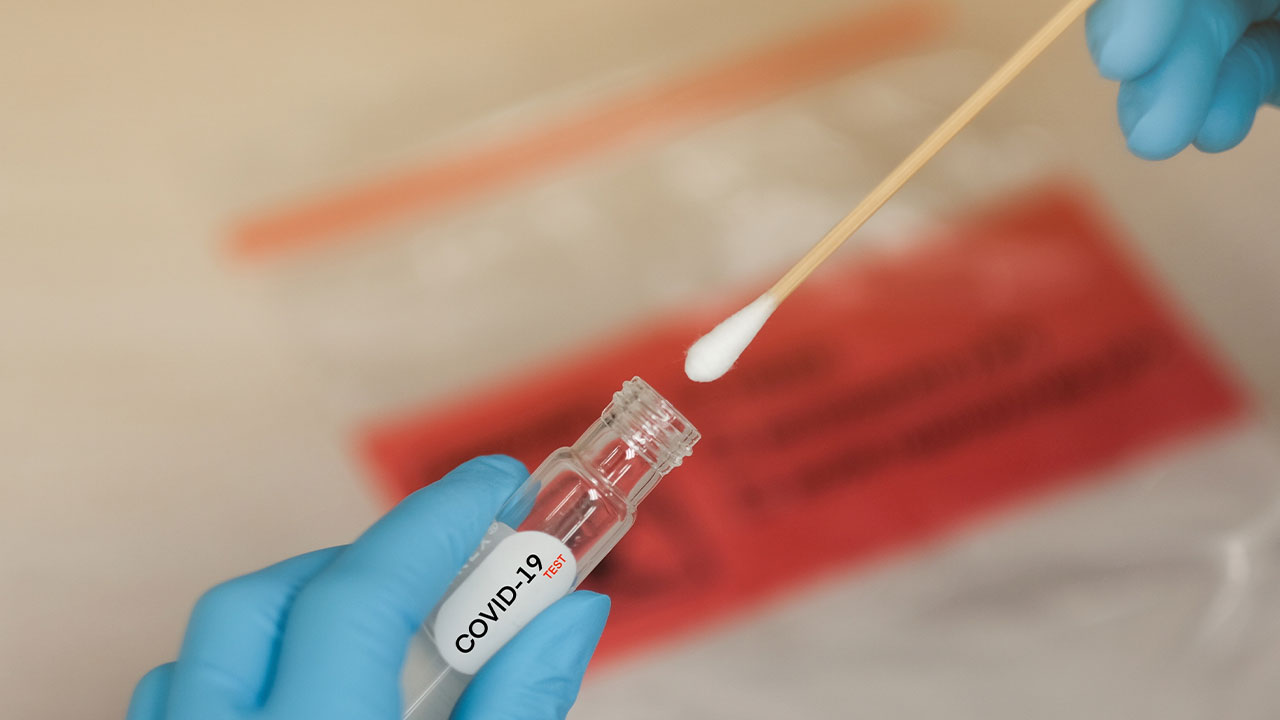
- TBG Diagnostics’ (TDL) subsidiary, TBG Biotechnology, has received U.S. Food and Drug Administration (FDA) approval for its COVID-19 antibody rapid test kits
- The FDA made the test kits available through the Emergency Use Authorisation (EUA)
- The test kit is immunochromatography based and uses colloidal gold technology to detect the presence of antibodies against N and S proteins of the SARS-CoV-2 virus
- The tests are manufactured by TBG Biotechnology in Taian and will be exported from Taiwan
- TBG Diagnostics shares remain suspended from official quotation, last traded for 27 cents each in March
TBG Diagnostics’ (TDL) subsidiary, TBG Biotechnology, has received United States Food and Drug Administration (FDA) approval for its COVID-19 antibody rapid test kits.
The test kit is immunochromatography based and uses colloidal gold technology to detect the presence of antibodies against N and S proteins of the SARS-CoV-2 virus.
The FDA made the test kits available through the Emergency Use Authorisation (EUA).
“The EUA is supported by the Secretary of Health and Human Service’s declaration that circumstances exist to justify the emergency use of in vitro diagnostics (IVDs)
for the detection and/or diagnosis of the virus that causes COVID-19,” the company explained.
As the test has been made available through the EUA, it has not undergone the same type of review as an FDA-approved or cleared IVD.
The tests are manufactured by TBG Biotechnology in Taian and will be exported from Taiwan.
“The Testing Kit is one of 41 in vitro diagnostics test kits for detecting antibodies against the novel coronavirus and one of 13 that uses the lateral flow immunochromatography rapid test platform that have received FDA EUAs to date,” the company added.
TBG Diagnostics shares remain suspended from official quotation, last traded for 27 cents each in March.